Project members

Project
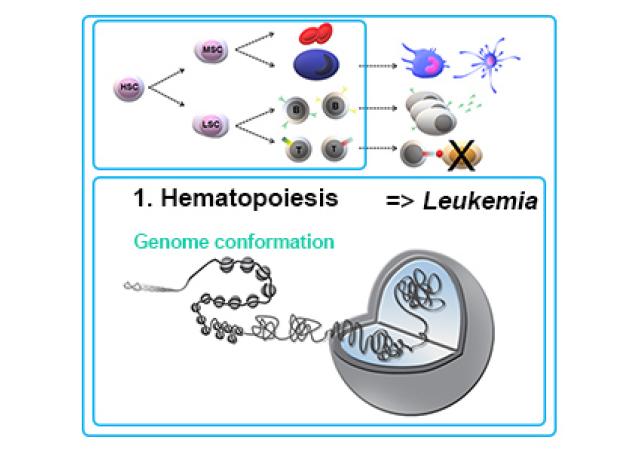
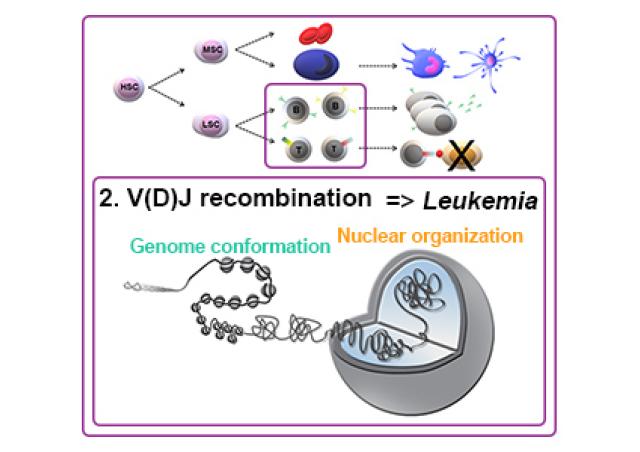
The project focuses on the regulation of the Tcra/d locus. Its recombination presents a unique challenge as: i) Tcrd is embedded within Tcra but they do not rearrange at the same developmental stage (Tcrd in early DN T cells, Tcra in later DP), ii) Tcra undergoes numerous recombination events in each DP, and iii) recombination can occur on both Tcra alleles. Finally, promiscuous non-functional recombination of the B-cell specific Igh locus in T cells adds to the complexity, and J. Chaumeil showed that dysregulation of Tcra/d and Igh recombination is linked to recurrent Tcra/d-Igh translocations underlying T-leukemia in ATM-deficient or RAG2-mutant cells (1). In the face of this complexity, maintaining genome integrity at the Tcra/d locus represents a major challenge and indeed, the pattern of the most common translocations in T-leukemia suggests so. Using genetic tools, we are directly assessing whether maintaining Tcra at different nuclear compartments, being repressive or permissive to transcription, can directly regulate its expression and rearrangement.
1. Proudhon C, Hao B, Raviram R, Chaumeil J, Skok JA. Long-Range Regulation of V(D)J Recombination. (2015) Adv Immunol, 128:123-82.
2. Chaumeil J, Micsinai M, Ntziachristos P, Roth DB, Aifantis I, Kluger Y, Deriano L, Skok JA. The RAG2 C-terminus and ATM protect genome integrity by controlling antigen receptor gene cleavage. (2013) Nature Comm, 31;4:2231.
3. Chaumeil J, Micsinai M, Ntziachristos P, Deriano L, Wang JMH, Ji Y, Nora E, Rodesch MJ, Jeddeloh JA, Kluger Y, Aifantis I, Schatz DG, Skok JA. Higher-order looping and nuclear organization of Tcra facilitate targeted RAG cleavage and regulated rearrangement in recombination centers. (2013) Cell Reports, 3(2):359-370.