Project members

Project
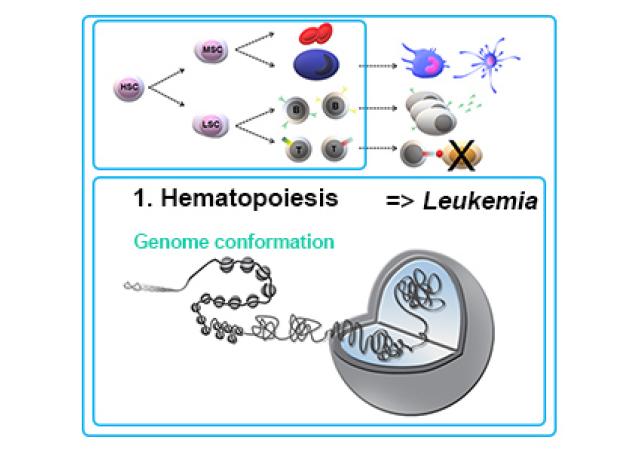
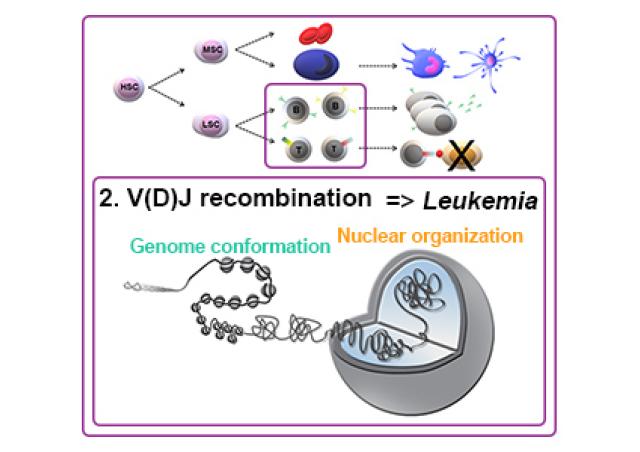
In collaboration with Dr JC Guéry (Infinity, Toulouse, France), we have demonstrated that the X-linked gene TLR7, coding a single-stranded RNA receptor, can escape XCI in a fraction of woman immune cells (1,2). Strikingly, B cells expressing TLR7 biallelically were more responsive than monoallelic cells at specific checkpoints of B cell differentiation that involve signaling through TLR7. Athough this overexpression of TLR7 gives to women a direct benefit with greater innate and adaptive immunity against selected pathogens or self-components containing ssRNA, it may also explain the higher susceptibility of women to TLR7-driven antibody mediated autoimmune diseases, such as SLE. We wish now to understand the mechanisms of TLR7-escape in immune cells.
- Souyris M, et al, Chaumeil J, Mejia JE, Guéry JC. Science Immunol 2018;3: pii: eaap8855.
- Souyris M, Mejia JE, Chaumeil J, Guéry JC. Semin Immunopathol 2018;41:153.