Rheumatoid arthritis (RA) is the most frequent inflammatory rheumatic disorder. It is a chronic autoimmune disease characterized by a chronic inflammation of the synovial tissue causing cartilage destruction, bone erosion, pain, and disability. Current RA therapies target the inflammatory consequences of autoimmune activation with the use of synthetic and biologic disease-modifying anti-rheumatic drugs. Despite the efficacy of agents, 30% of patients seem to have no response or no sustained response. This primary or secondary lack of clinical response upon treatments targeting inflammation supports the need to develop new complementary therapeutic strategies.
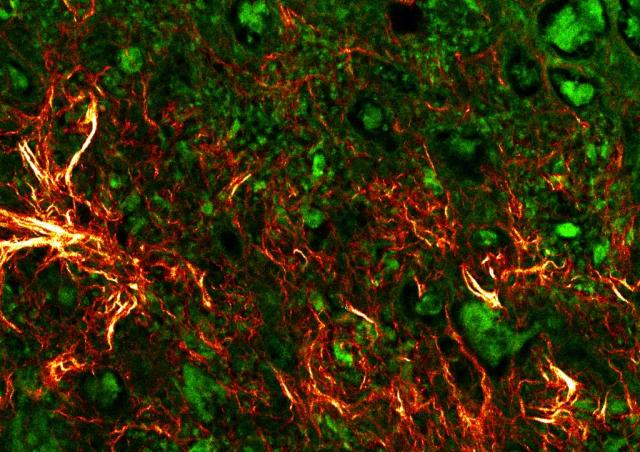
The synovium is the primary site of the inflammatory process, which if untreated leads to irreversible damage to the adjacent cartilage and bone. Inflammatory infiltrates and formation of new blood vessels are crucial events to promote the development of the hyperplasic proliferative pathologic synovium. Our objective is to identify new actors of synovial inflammation and angiogenesis with the ambition to develop new relevant biomarkers of disease activity or severity as well as new therapeutic strategies.
Project members
Project
Our objective is to identify new actors of synovial inflammation and angiogenesis. The expression of identified candidates is then assessed in endothelial cells, synovial cells, synovial fluid, serum and synovial tissue. We have established for this purpose a huge databank of biologic samples. The expression or activity of identified candidates is then modulated by complementary approaches to study the functional properties of endothelial cells, immune cells or synoviocytes. The effects of the modulation of target expression or activity are then studied in vivo in mouse models of neoangiogenesis and experimental arthritis. We are working thanks to the expertise and recruitment at Cochin University Hospital.