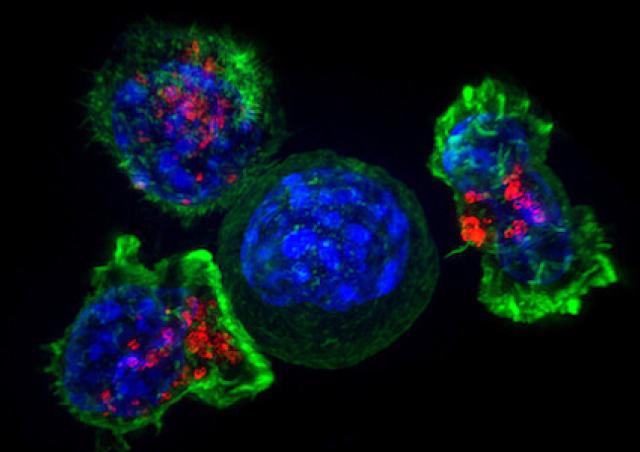
Members of the project
Project
The ability of the immune system (IS) to respond to tumor antigens has naturally led to the development of therapeutic trials in humans. In the 1990s, the first trials of immunotherapy, such as the use of IL-2 or IFN-α, were not particularly encouraging due to mitigated results and collateral toxicities. However, we know from these early therapeutic trials that the tolerogenic environment of tumors is dominant and prevents IS from eliminating them. One of the many discovered mechanisms of tumor escape is the induction of an inability of effector T lymphocytes (TLs) to respond to a tumor antigen, a mechanism called "exhaustion". This mechanism has since become the target of therapeutic trials. There are, in fact, control points of the IS, called "Immune Checkpoints", corresponding, among others, to the expression by TLs of inhibitory molecules that block their effector functions. The inhibition of these checkpoints allows the restoration of effector TLs function.
Revolutionising the treatment of many cancers such as melanoma, these new immunotherapies (anti-CTLA-4 and anti-PD-1 in particular) increase the survival of patients. Unfortunately, only 20-30% of patients respond successfully to these treatments by developing an effective anti-tumor immune response. To overcome this insufficiency, current strategies propose to combine immunotherapy with the more traditional treatments such as chemotherapy and radiotherapy.
Our first results show that iron significantly increases TLs responses in vivo and in vitro. This "adjuvant" effect results in a strong slowing down of tumor cell growth after transplantation in mice. Therefore, we propose to enhance the efficacy of immunotherapy by augmenting and shaping LT response with one of the most classical chemical elements, iron.

Aim #1: Iron supplementation, IFN-g production by TLs and ferroptosis of tumor cells
Our preliminary results show that iron supplementation results in a significant slowing of tumor growth in mice tumor transplantation models. In this context, iron promotes the differentiation/expansion of type 1 (Th1) effector TLs, which are characterised by the expression of the transcription factor T-bet and by their ability to produce high amounts of IFN-γ. Finally, using an antigen-specific system, we observed, in vitro, that iron strongly increased the differentiation capacity of naive CD4 TLs into Th1-type cells. We hypothesise that iron, by increasing the response of anti-tumor TLs and in particular their capacity to produce IFN-g, would lead to cancer cell death by induction of ferroptosis. Ferroptosis is a recently described atypical cell death mechanism involving the destruction of cell membranes by oxidative stress following the failure of a glutathione-dependent antioxidant system. Ferroptosis can be induced by increasing intracellular iron levels or by inactivating glutathione peroxidase 4 (GPX4). We therefore propose that it is through the induction of IFN-γ production by TLs that iron supplementation would exert its anti-tumor activity in vivo. Indeed, it has recently been suggested that IFN-γ may inhibit GPX4 by limiting Cystine uptake by cells. We therefore wish to test this hypothesis using tumor cell lines genetically modified with CRISPR/Cas9 technology to render them insensitive to either ferroptosis or IFN-γ.

Aim #2: Iron and anti-PD1 immunotherapy
Our results show a beneficial effect of iron supplementation on anti-tumor responses for the different tumor cell lines we have tested. We now plan to test the effect of iron supplementation and anti-PD1 antibody injection combination (antibody blocking the interaction between PD1 and its ligand PDL1) on tumor growth.
Each treatment, separately, increases anti-tumor immune responses, leading to a slowing down or even arrest of tumor growth. We expect that the effects of these two treatments will not only be additive, but also that a synergy can be observed, i.e. that the anti-tumor effect of the two treatments together is greater than the sum of the effects of the two treatments applied separately. We will test tumor cell lines known to be "responsive" to anti-PD1 immunotherapy as well as tumor cell lines that are rather "resistant" to the same treatment. Iron supplementation should increase immune response against responder lineages induced by anti-PD1 and regression or even complete elimination of tumors should be observed. In the case of "resistant" cell lines, we will verify whether iron supplementation will allow transplanted mice to respond to anti-PD1 treatment and thus control the growth of these tumors. To make a homology with patients, we hope that individuals normally non-responders to immunotherapy become responders when supplemented with iron.