Project members
Project
Stimulating endogenous craniofacial regeneration is necessary to treat severe tissue damage and avoid the need for bone transplantation together with its potential clinical complications. Thus, it is essential to be able to promote a controlled cellular dedifferentiation to increase cell plasticity and enhance craniofacial tissue repair.
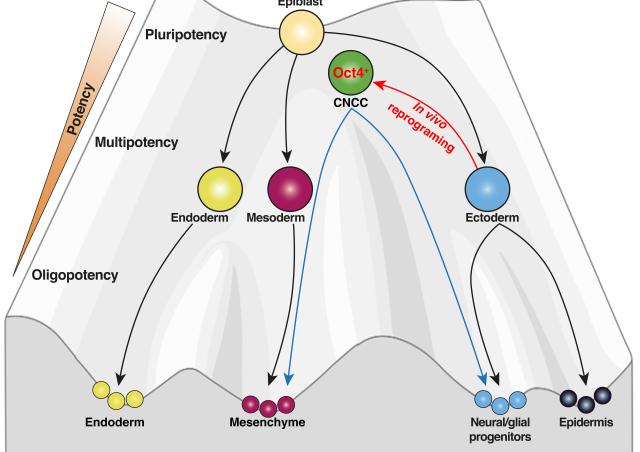
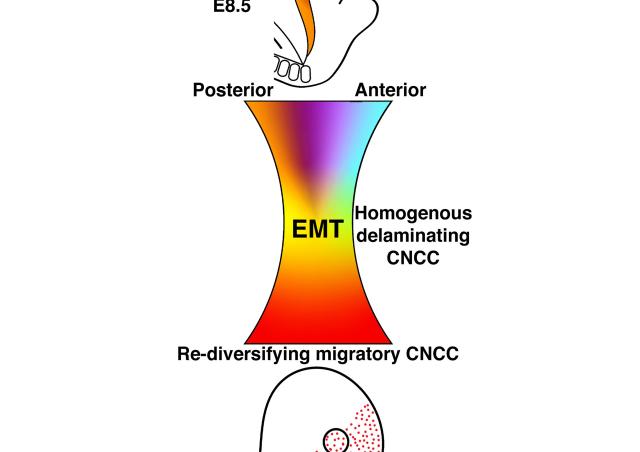
Most craniofacial bones are derived from cranial neural crest cells, a population that transiently returns into pluripotency during development. My preliminary observations suggest cranial neural crest cell derivatives retain the potential to reverse cell differentiation during post-natal mandibular regeneration.
The transcriptional regulatory mechanisms stimulating this process during lower jaw regeneration remain unknown.

To decipher the mechanisms that could enhance mandibular regeneration we are characterizing the spatiotemporal origin of the cells reactivating pluripotency programs during tissue repair using multi-photon in vivo imaging. We are also characterizing their molecular identities with transcriptomics and chromatin accessibility methods.
Together, this work will elucidate the molecular mechanisms able to stimulate mandibular regeneration and open new strategies to manipulate these programs to improve craniofacial tissue repair.