In the 1990s, Professor Jean-Philippe Wolf worked in the Medically Assisted Reproduction laboratory at the Kremlin Bicêtre Hospital, then at the Cochin Hospital in Paris and collaborated with G. Palermo from Professor Van Steirteghem's department, in Brussels. The aim was to develop techniques for micro-injecting sperm into eggs to improve fertilization rates in couples whose partner had particularly deficient sperm.
The first technique considered consisted of microinjecting the spermatozoa under the Pellucidal Zone which surrounds the oocyte (Figure 1). This technique was called SUZI for “Sub Zonal Insemination”. G. Palermo, during such an SUZI attempt, made the sperm enter directly inside the egg and thus created ICSI, which everyone knows, and which allowed the birth of many children.
Jean-Philippe Wolf's research group was then interested in the identification of proteins involved in the interaction between gametes, allowing the recognition, adhesion and fusion of gametic membranes. At that time, P. Primakoff's group at UCLA had just published an article in Cell in which it suggested that the sperm receptor on the membrane of the mouse oocyte was the integrin α6β1.
The team wanted, firstly, to check if this was also the case in the human species. But instead of classically using antibodies against the α6 or β1 integrin subunit, Morgane Bomsel from Institut Cochin, also involved in this project, had the idea of synthesizing a peptide reproducing the presumptive sperm ligand. If the receptor was the integrin α6β1, the ligand must have been the disintegrin domain of the ADAM2 protein, otherwise called Fertilin β. A peptide reproducing its binding site, with a few modifications, the FEE peptide, was therefore synthesized.
FEE peptide increases fertilization rate, in vitro
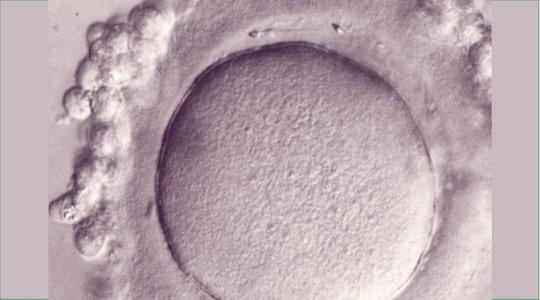
The only model on which the peptide could be tested in the human species was the model of the immature oocyte blocked at the Germinal Vesicle (VG) stage, and given for research. Indeed, these oocytes cannot be used for the couple's child project, their meiosis is blocked at the 1st stage.
These oocytes had to be depellucidated (fig. 1a) to avoid creating an embryo, which was prohibited in the context of research. Around twenty spermatozoa can then fuse with the depellucidated oocyte, since there is no longer the blockage of polyspermy normally located at the Pellucidal Zone.
Such oocytes were therefore inseminated with or without the FEE peptide with the aim of achieving a dose-dependent inhibition of fertilization with the molecule. The fused spermatozoa were fluorescently labeled (Hoechst). The researchers then had the enormous surprise to note that contrary to what was expected, the molecule did not inhibit the passage of sperm inside the egg but on the contrary increased their number (Figure 2b and c) (Ziyyat et al., Cyclic FEE peptide increases human gamete fusion and potentiates its RGD-induced inhibition. Hum Reprod. 2005).

The discovery was surprising but nonetheless very interesting since it made it possible to improve fertilization rates during In Vitro Fertilization (IVF), which represented a significant technological leap and responded to a societal challenge. The “Assistance Publique” and the University have therefore taken out a Patent on the molecule and a request for Clinical Trial authorization has been filed with the Biomedicine Agency.
The FEE peptide from murine Fertiline increases the fertilization rate in vivo in mice
To obtain Clinical Trial authorization, in vivo studies were carried out in mice. It was thus shown, on intact cumulo-oocyte complexes (mature oocytes surrounded by follicle cells, Figure 1), that the homologous peptide derived from murine Fertiline, had the same properties as the human peptide, and above all that it allowed to obtain healthy offspring that reproduce normally over 3 generations (Barraud-Lange et al., Cyclic QDE peptide increases fertilization rates and provides healthy pups in mouse. Fertil Steril. 2009).
FEE peptide enhances human egg maturation and chromosome segregation during meiosis I
The properties of the peptide on the maturation of human oocytes were then studied in more detail. The model used was that of the in vitro maturation of oocytes blocked at the VG stage, but with the Pellucidal Zone intact this time. More than 1,600 oocytes were included in the study, which showed that the FEE peptide improved their maturation rate by 30% compared to the control group. As part of a collaboration with the Department of Professor Serge Romana (Necker Hospital), a chromosomal analysis technique using DNA chips (CGH array) was developed on a single cell, to establish the chromosomal formula of thus matured oocytes.
To give birth to a child after fertilization, an oocyte must have completed meiosis and above all be euploid (i.e. have the normal number of chromosomes), this property decreasing significantly with the age of women, which is the cause of the decline in their fertility. Surprisingly, the FEE peptide also improved chromosome segregation during meiosis I and the euploidy rate of matured oocytes at the M II stage (Sallem et al., A fertilin-derived peptide improves in vitro maturation and ploidy of human oocytes. F S Sci. 2022).
FEE peptide improves in vitro Pre-implantation development of human embryos and hatching of blastocysts
JP Wolf's group then obtained authorization for “research on human embryos” from the “Agence de la Biomédecine”. These are embryos obtained for couples who, no longer having parental plans, donate them to research.
After verifying that the peptide effectively marked the blastomeres of J2 embryos at their interface, a dose-response study of blastoformation over 4 days was carried out. This work thus demonstrated a dose-dependent stimulation of the blastoformation of these embryos by the FEE peptide, and interestingly an increase in the number of trophectoderm cells (future placenta) without modification of the embryonic button (which will give rise to the baby). Interestingly, the number of trophectoderm cells is the variable most correlated with the percentage of live births. In addition, the spontaneous hatching rate of these embryos was multiplied by 3, thus demonstrating their evolutionary capacity (Denizot et al., Cyclic fertilin-derived peptide stimulates in vitro human embryo development. F S Sci. 2022 Feb;3(1):49-63).
IVF clinical trials with FEE peptide
A first clinical trial began in 2015, but could only last 3 months for administrative reasons (anomaly during the revision of the Bioethics law, etc.). During this first trial, 66 couples were included for IVF, their oocytes being randomly distributed into two groups, and inseminated with the spouse's sperm in the presence or absence of the FEE molecule. The percentage of births increased by 7% in the treated group compared to the control group, and this percentage rose to 13% during the first transfer carried out with the embryo from the cohort with the best prognosis.
A phase 3 clinical trial of which AP-HP is Promoter is currently underway. It will include 360 couples divided into two groups, in 8 care establishments practicing Medically Assisted Reproduction. The babies will be followed for a year. Results in 2025… With the key being a request for marketing authorization.