Genome stability and instability (SInG)
Genome stability maintenance is essential to prevent aging, cancer, neuronal and/or congenital disorders. The organisms are continually challenged by stresses (radiation, chemical contaminants, free radicals ...) that damage the DNA and thus that generate genetic instability. Our laboratory is interested in the subtle balances of between the different mechanisms that orchestrate DNA repair, protecting against genetic instability while allowing genetic diversity (antibodies, gametes, molecular evolution).
The integrity of the genetic material is daily compromised by exogenous (radiation exposure) or endogenous attacks. Endogenous stress can result from reactive oxygen species (ROS) generated by cell metabolism; in addition, replicative stresses also represent important endogenous stressors. Indeed, the progression of the replication forks is inevitably blocked by numerous endogenous structural damages or configurations (DNA / RNA hybrids, DNA structure difficult to replicate, strong protein binding, etc.). It can be noted that oxidative stress and replicative stress are generally considered both tumorigenic and promoting senescence that counteracts tumour development. The transmission of the genetic information therefore requires the control of the DNA damage response (DDR), which coordinates a network of metabolic pathways. It should be emphasized that spontaneous activation of DDR, in response to endogenous replicative stress, has been described in precancerous cells or at early stages of tumour initiation. In addition, DDR defects are also associated with developmental, neuronal or inflammation pathologies. Thus the study of the nature and consequences of endogenous replicative stress is of crucial importance for the understanding of spontaneous genetic instability.
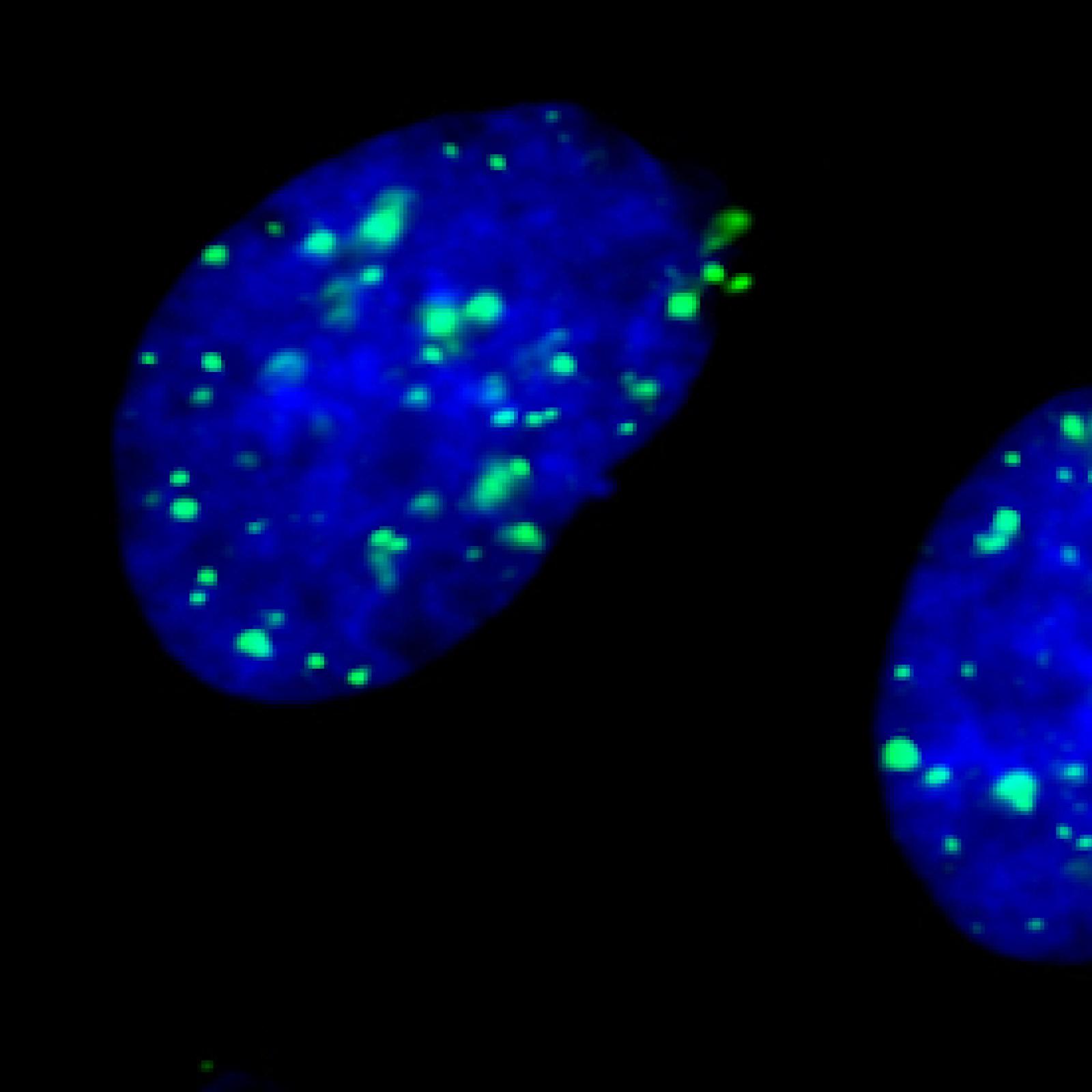
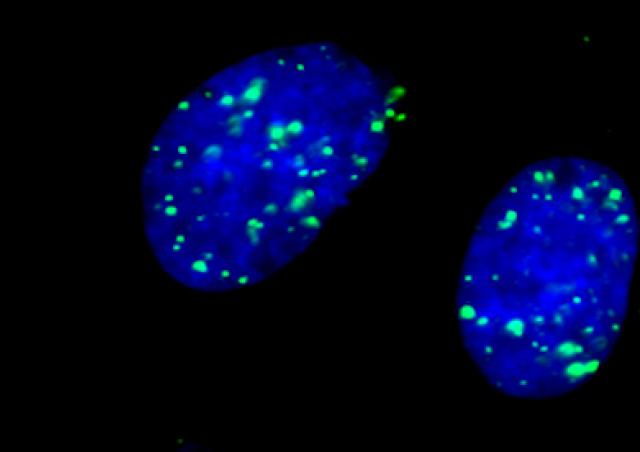
DNA Double-strand breaks (DSBs) are highly toxic lesions that can be produced by ionizing radiation or arrests of the replication forks. DSBs can also be physiologically produced by the cell during meiotic differentiation or V (D) J recombination generating the immune repertoire. The study of CBD repair therefore has both fundamental and applied interests. Indeed, on the one hand, strategies using radiotherapy should largely benefit from knowledge on the regulation of DSB repair. On the other hand, if DSBs can be used to generate genetic diversity (meiosis, V (D) J recombination) they can also create genetic instability. Fine and precise regulation of DSB repair is therefore essential to preserve these balances and cellular issues.
The metabolic network repairing DNA damages must therefore support the maintenance of genome stability, while allowing genetic diversity. For some lesions, several alternative mechanisms can act; it is therefore essential to coordinate the different repair systems between them, but also with other fundamental processes of the cell, such as DNA replication, the cell cycle and apoptosis.
DSB repair uses two main general strategies:
- The first strategy uses one DNA partner sharing homologous and refers to homologous recombination (RH). This is a process, conserved in all organisms, and is at the heart of the balance maintaining the balance between stability and genetic diversity. In addition to DSBs repair, RH allows the restart of blocked replication forks, which gives it a protective role against endogenous replicative stress.
- The second DSB repair strategy ligates the double-stranded ends without requiring sequence homologies (NHEJ: Non-homologous End-joining).